Oxide of high porosity (>50%) is found to deposit in drum as well as once-through boilers under both low and high oxygen water chemistry conditions. The deposition rate is approx. proportional to the concentration of particulate iron oxide and the square of the heat flux. The best approximation to the real situation is given by
D = k q2 c t
Where,
D = amount of magnetite deposited (kg/m2)
q = the heat flux (W/m2)
c = concentration of iron in water (kg/m3)
t = time (hour)
k = constant ( approx. 5 X 10-13 / W2 m2/s
In a wick boiling mechanism, salts dissolved in the boiler water can be concentrated by factors > 104 as shown in the figure below.
Generally, the protective magnetite scale thickness is 10-15 microns in the waterwall tube. When the corrosion rate increases due to upset of water chemistry parameters in boiler, (due to salt ingress and concentration), the deposit formation also increases due to corrosion of metal and precipitation of contaminants whose water solubility decreases at higher temperature on the evaporator tube surface. To maintain the pH in boiler water, in case of reduction of pH due to salt ingress, addition of more Tri Sodium Phosphate (TSP) is required. In this process, at some places on the internal surface of waterwall tubes, deposit thickness increases and the protective iron oxide scale becomes non protective and porous in nature. Porous, insulating types of deposits allow boiler water to diffuse into the deposit where the water becomes trapped and boils.
The boiling of deposit in entrapped water produces relatively pure steam which tends to diffuse out of the deposit, leaving behind super heated non-boiling equilibrium solution of caustic, which is responsible for caustic corrosion or acidic solution, which is responsible of hydrogen damage in waterwall tubes as discussed below.
IRC is regularly carrying out such studies to find out the reason of the failures and are also giving remedial actions to prevent it, if required we can also carry out chemical cleaning of boiler.
IRC is a service provider having expertise in ndt, residual life assessment, fitness for service, advanced ndt, failure investigation, chemical cleaning, certification of storage tanks as per chief controller of explosives guidelines , consultancy for boiler water chemistry and training
 IRC Engineering Services
IRC Engineering Services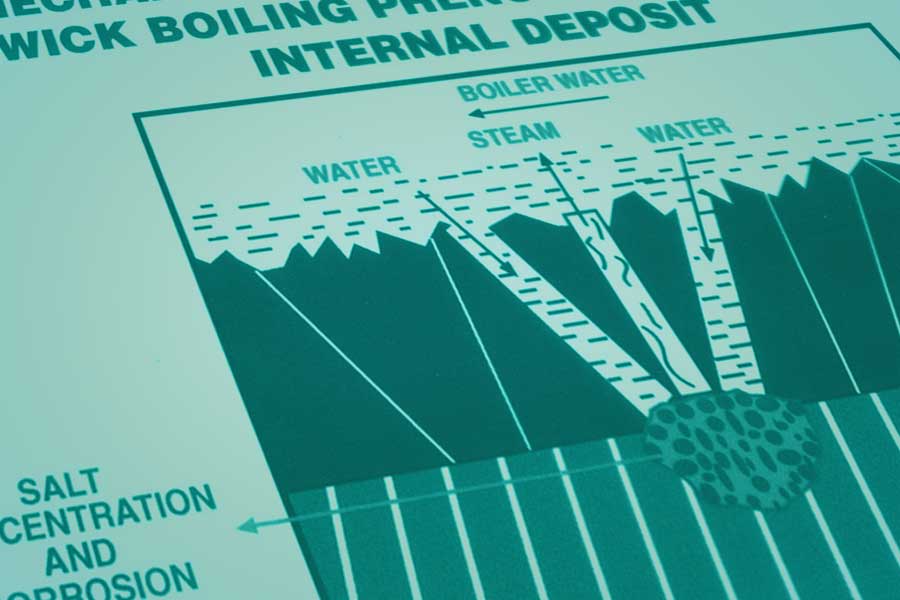


 Photo 1
Photo 1  Photo 2
Photo 2  Photo 3
Photo 3  Photo 4
Photo 4 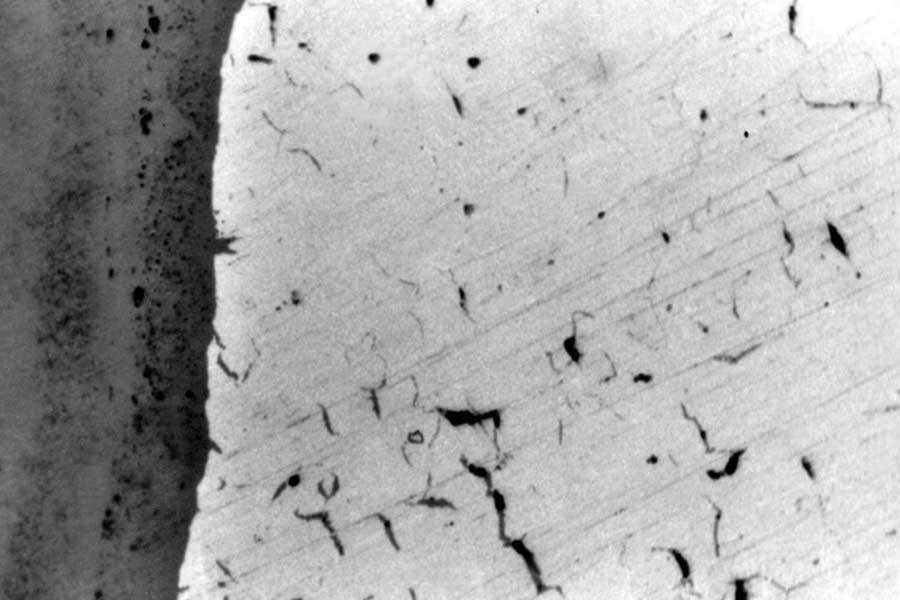







Recent Comments